UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): April 8, 2026
(Exact name of registrant as specified in its Charter)
Not applicable | |||||||||||
(State or other jurisdiction of incorporation or organization) | (Commission File Number) | (I.R.S. Employer Identification No.) | |||||||||
(Address of principal executive offices)(Zip Code) | |||||||||||
| (Registrant's telephone number, including area code) | |||||||||||
Not Applicable | |||||||||||
(Former name or former address, if changed since last report) | |||||||||||
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | |||||
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | |||||
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | |||||
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) | |||||
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||||||
* | Not for trading, but only in connection with the listing of the American Depositary Shares on The Nasdaq Global Select Market. The American Depositary Shares represent the right to receive ordinary shares and are being registered under the Securities Act of 1933, as amended, pursuant to a separate Registration Statement on Form F-6. Accordingly, the American Depositary Shares are exempt from the operation of Section 12(a) of the Securities Exchange Act of 1934, as amended, pursuant to Rule 12a-8 thereunder. | |||||||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 7.01 Regulation FD Disclosure.
On April 8, 2026, Autolus Therapeutics plc (the “Company”) hosted a virtual investor event entitled “Spotlight on the Acute Lymphoblastic Leukemia (ALL) Business” and posted the presentation materials to its website. A copy of the presentation is attached hereto as Exhibit 99.1 and is incorporated herein by reference.
The information furnished under this Item 7.01, including Exhibit 99.1, shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, or the Exchange Act, or subject to the liabilities of that section or Sections 11 and 12(a)(2) of the Securities Act of 1933, or the Securities Act. The information in this Item 7.01, including Exhibit 99.1, shall not be deemed incorporated by reference into any other filing with the U.S. Securities Exchange Commission, or the SEC, made by the Company, whether made before or after the date hereof, regardless of any general incorporation language in such filing.
Item 9.01 Financial Statements and Exhibits
d) Exhibits
| Exhibit No. | Description of Exhibit | |||||||
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) | |||||||
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| AUTOLUS THERAPEUTICS PLC | ||||||||
Dated: April 8, 2026 | By: | /s/Christian Itin, Ph.D. | ||||||
| Name: Christian Itin, Ph.D. | ||||||||
| Title: Chief Executive Officer | ||||||||

Spotlight on the Acute Lymphoblastic Leukemia (ALL) Business AUTOLUS INVESTOR EVENT April 8, 2026 EX-99.1

Disclaimer These slides contain forward-looking statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995. Forward-looking statements are statements that are not historical facts, and in some cases can be identified by terms such as “may,” “will,” “could,” “expects,” “plans,” “anticipates,” and “believes.” These statements include, but are not limited to: statements regarding Autolus’ development and commercialization of its product candidates; Autolus' manufacturing, sales and marketing plans for AUCATZYL, including expectations regarding the commercial launch in the United States and the ability to reach patients in a timely manner; the amount and timing of milestone payments under Autolus' collaboration and license agreements; and future development plans of obe-cel, including the timing or likelihood of expansion into additional markets or geographies and related regulatory approvals. Any forward-looking statements are based on management’s current views and assumptions and involve risks and uncertainties that could cause actual results, performance, or events to differ materially from those expressed or implied in such statements. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of various risks and uncertainties, which include, without limitation: Autolus' ability to maintain regulatory approval of AUCATZYL; its ability to execute its commercialization strategy for AUCATZYL; its ability to develop, manufacture and commercialize its other product candidates and the timing or likelihood of expansion of AUCATZYL into additional markets or geographies; Autolus' ability to establish and expand a commercial infrastructure and to successfully launch, market and sell AUCATZYL; actions of regulatory agencies, which may affect the initiation, timing and progress of clinical trials or future regulatory approval; the labelling for AUCATZYL/obe-cel in any future indication or patient population, if approved; the potential for payors to delay, limit or deny coverage for AUCATZYL; Autolus' ability to obtain, maintain and enforce intellectual property protection for AUCATZYL or any product candidates it is developing; the results of clinical trials are not always being predictive of future results; the cost, timing and results of clinical trials; that many product candidates do not become approved drugs on a timely or cost effective basis or at all; the ability to enroll patients in clinical trials; and possible safety and efficacy concerns. For a discussion of other risks and uncertainties, and other important factors, any of which could cause Autolus’ actual results to differ from those contained in the forward-looking statements, see the section titled “Risk Factors” in Autolus' Annual Report on Form 10-K filed with the Securities and Exchange Commission, or the SEC, on March 27, 2026, as well as discussions of potential risks, uncertainties, and other important factors in Autolus’ subsequent filings with the Securities and Exchange Commission. All information in this presentation is as of the date of the presentation, and Autolus undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events, or otherwise, except as required by law. You should, therefore, not rely on these forward-looking statements as representing the Company’s views as of any date subsequent to the date of this presentation. 2 Investor Event: Spotlight on ALL Business

Important Safety Information 3 • AUCATZYL indicated for the treatment of adults with relapsed or refractory B-cell precursor acute lymphoblastic leukemia (B-ALL) • The safety of AUCATZYL includes a boxed warning for CRS, neurologic toxicities, and secondary hematological malignancies. ICANS, including fatal or life- threatening reactions, occurred in patients receiving AUCATZYL. T-cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T-cell immunotherapies. • In the FELIX trial, severe, including life-threatening and fatal infections occurred in patients after AUCATZYL infusion. The non-COVID-19 infections of all grades occurred in 67% (67/100) of patients. Grade 3 or higher non-COVID-19 infections occurred in 41% (41/100) of patients. • Please see full Prescribing Information, including BOXED WARNING and Medication Guide. Investor Event: Spotlight on ALL Business

Spotlight on the ALL Business Agenda – April 8, 2026; 1:00 – 2:15pm EDT Investor Event: Spotlight on ALL Business 4 Jae Park, Lori Muffly, Elias Jabbour and Michael Pulsipher are paid consultants of Autolus Therapeutics. Jae H. Park, MD Lori Muffly, MD, MS Elias Jabbour, MD Michael Pulsipher, MD Memorial Sloan Kettering Cancer Center Stanford School of Medicine MD Anderson Cancer Center University of Utah Huntsman Cancer Institute Welcome and introduction of speakers Dr. Matthias Will Adult ALL treatment landscape & unmet medical need Dr. Jae Park ROCCA real world experience with AUCATZYL® Dr. Lori Muffly IST focus on exploring obe-cel as definitive frontline consolidation Dr. Elias Jabbour Pediatric medical need and Autolus’ recent CATULUS data Dr. Michael Pulsipher Q&A All ALL business opportunity and outlook Dr. Christian Itin

Memorial Sloan Kettering Cancer Center A U T O L U S I N V E S T O R S E R I E S · 2 0 2 6 Unmet Needs in Relapsed / Refractory Adult B-ALL Independent Perspectives on the Present and Future of the Field Jae H. Park, MD Director, Adult ALL Program | Acting Chief, Cellular Therapeutics Memorial Sloan Kettering Cancer Center · New York, NY The views expressed represent the independent scientific perspective of Dr. Park and are not intended as promotional statements regarding any specific product. Dr. Park discloses a consulting relationship with Autolus Therapeutics.

Memorial Sloan Kettering Cancer Center Department of Medicine | Leukemia Service Adult R/R B-ALL: A Disease Where Standard Approaches Fall Short ~1,800 New R/R ALL cases annually in the US <40% 5-year OS with standard salvage ~50% Patients ineligible for allo-SCT ≥2 Lines Average prior therapy before CAR-T access The clinical reality: Adult R/R B-ALL carries a fundamentally different biology and risk profile than pediatric disease. Median OS after first relapse is approximately 6–9 months with chemotherapy- and non-CAR immunotherapy-based salvage. Even for patients who achieve CR2, sustained remission without transplant is rare. The field urgently needs therapies that are durable without requiring SCT — particularly for transplant-ineligible patients due to age, comorbidity, or prior treatment. Sources: SEER data; Gökbuget et al., Blood 2012; Roddie et al., NEJM 2025

Memorial Sloan Kettering Cancer Center Department of Medicine | Leukemia Service The R/R ALL Treatment Pathway: A Cascade of Attrition Adult R/R B-ALL at 1st Relapse ~1,800 pts/yr 100% Achieve CR2 with Blinatumomab/Salvage ~990 pts ~55% Eligible & Proceeds to Allo-SCT ~450 pts ~25% Receives CAR-T (currently) ~270 pts ~15% No standard curative option ~810 pts ~45% Where the unmet need concentrates Transplant-ineligible patients (largest gap) ~45-50% of R/R adults cannot proceed to allo-SCT. For these patients, there is no curative standard of care. CAR-T is the only approach with potential for durable remission without transplant. Post-relapse after blinatumomab Outcomes after blinatumomab failure are dismal — ORR with subsequent chemotherapy is <30%. CAR-T in this population represents a critical salvage option, and sequencing data are emerging. Ph+ ALL after TKI failure Despite ponatinib combinations improving frontline outcomes, Ph+ ALL patients who relapse on TKI therapy have extremely limited options. CD19 CAR-T efficacy appears maintained regardless of Ph status. CNS disease involvement CNS relapse and extramedullary disease in B-ALL carry particularly poor prognosis with conventional agents. Early CAR-T data suggest CNS penetration and activity, but this population remains underrepresented in trials. Source: Park et al., NEJM 2025; Gökbuget et al.; NCCN B-ALL Guidelines v2.2025

Memorial Sloan Kettering Cancer Center Department of Medicine | Leukemia Service CAR-T Therapy: Improving Outcomes in Adult B-ALL FELIX Study 77% Overall response rate across all patients >3 yrs Durable remissions in low disease burden cohort MRD Strong predictor of long-term OS after CAR-T CAT-19 Differentiated CAT-19 binder with intermediate affinity due to a fast binding off rate What the data tell us about the future 1 Disease burden at infusion is the critical variable Patients with low disease burden have dramatically improved outcomes — both efficacy and toxicity. This argues strongly for earlier deployment of CAR-T, before extensive salvage therapy exhausts the patient. 2 CAR-T can be a bridge to transplant — or a bridge away from it For patients who achieve deep MRD-negative remission, the data support durable remission without SCT. This is a paradigm shift for transplant-ineligible patients. 3 Product phenotype matters as much as cell dose Memory T cell content, exhaustion markers, Tscm/Tcm ratio — these are independent predictors of durability. Manufacturing science is now a direct clinical differentiator. Source: Roddie et al., New England Journal of Medicine, 2024; Park JH et al. EHA 2025; Simpson et al. ASH Annual Meeting 2025; MSK Phase I 19-28z trial (NCT01044069)
Memorial Sloan Kettering Cancer Center Department of Medicine | Leukemia Service Current Limitations/Aucatzyl Solutions 01 SafetyToxicity profile in heavily pretreated adults Older adults with comorbidities have poor tolerance of severe CRS and ICANS. Current approved products carry meaningful Grade 3+ toxicity rates that limit eligibility — particularly in community settings. A safer toxicity profile determines who can access therapy. 02 ManufacturingCAR-T product fitness and manufacturing reliability T cells from heavily pretreated, lymphopenic ALL patients are challenging to manufacture. Vein-to- vein time, product failure rates, and infusion product phenotype vary significantly across approved products. Consistency of manufacturing is a clinical outcome variable. 03 ResponseLong-term persistence and resistance mechanisms Persistency testing is now available for Aucaztyl. CD19 antigen loss or downregulation is observed in 20-30% of relapses post-CAR-T. 04 AccessAccess, vein-to-vein time, and operational barriers Median vein-to-vein times of 28–56 days create a critical window where patients may deteriorate before receiving therapy. Shortening this timeline — through process innovation and manufacturing efficiency — is a direct patient outcome intervention. Independent clinical assessment — Jae H. Park, MD | Memorial Sloan Kettering Cancer Center

I N D E P E N D E N T P E R S P E C T I V E Where Is the Field Heading? An Honest Assessment. CAR-T will move earlier in the treatment algorithm The strongest predictor of durable response is low disease burden at the time of infusion. The data now support investigating CAR-T in the CR1 or MRD-positive setting in earlier line treatment — not just as a last resort. Product biology will become the competitive differentiator As more CD19 CAR-T products enter the market, differentiation will come from product phenotype — memory enrichment, persistence, tonic signaling control. Constructs optimized for T cell fitness will outperform older designs. Manufacturing speed and reliability are clinical outcomes Vein-to-vein time is a survival variable in ALL. Products that reliably deliver a fit product in <30 days will have a meaningful clinical and commercial advantage. This is a medical issue, not a supply chain issue. Combination strategies will define the next generation Single-agent CD19 CAR-T will be surpassed by rational combinations — dual antigen targeting to prevent escape, checkpoint blockade to sustain persistence, or armored CAR-T to overcome the immunosuppressive TME. These perspectives reflect Dr. Park's independent scientific views based on published evidence and clinical experience. Dr. Park discloses a consulting relationship with Autolus Therapeutics.
Memorial Sloan Kettering Cancer Center Department of Medicine | Leukemia Service Defining Success: What the Field Needs to Deliver for Patients "The goal for the next decade in adult ALL is straightforward but demanding: a therapy potent enough to produce deep MRD-negative remission, safe enough to be administered broadly including in the elderly and those with comorbidities, fast enough that patients don't deteriorate waiting for their product, and durable enough that transplant becomes optional rather than mandatory for appropriate patients." — Jae H. Park, MD | MSK 1 Durable MRD-negative remission without mandatory SCT Redefining cure as accessible to transplant-ineligible patients 2 Fit-for-purpose T cell products Manufacturing science that preserves memory phenotype and minimizes exhaustion 3 Reduced toxicity enabling earlier use CRS/ICANS profiles manageable in community settings and in older adults Jae H. Park, MD | Memorial Sloan Kettering Cancer Center | Autolus Investor Call 2026 | 1275 York Avenue, New York, NY 10065

ROCCA Real World Experience in AUCATZYL® First Year Lori Muffly, MD, MS PROFESSOR OF MEDICINE BLOOD AND MARROW TRANSPLANTATION AND CELLULAR THERAPY
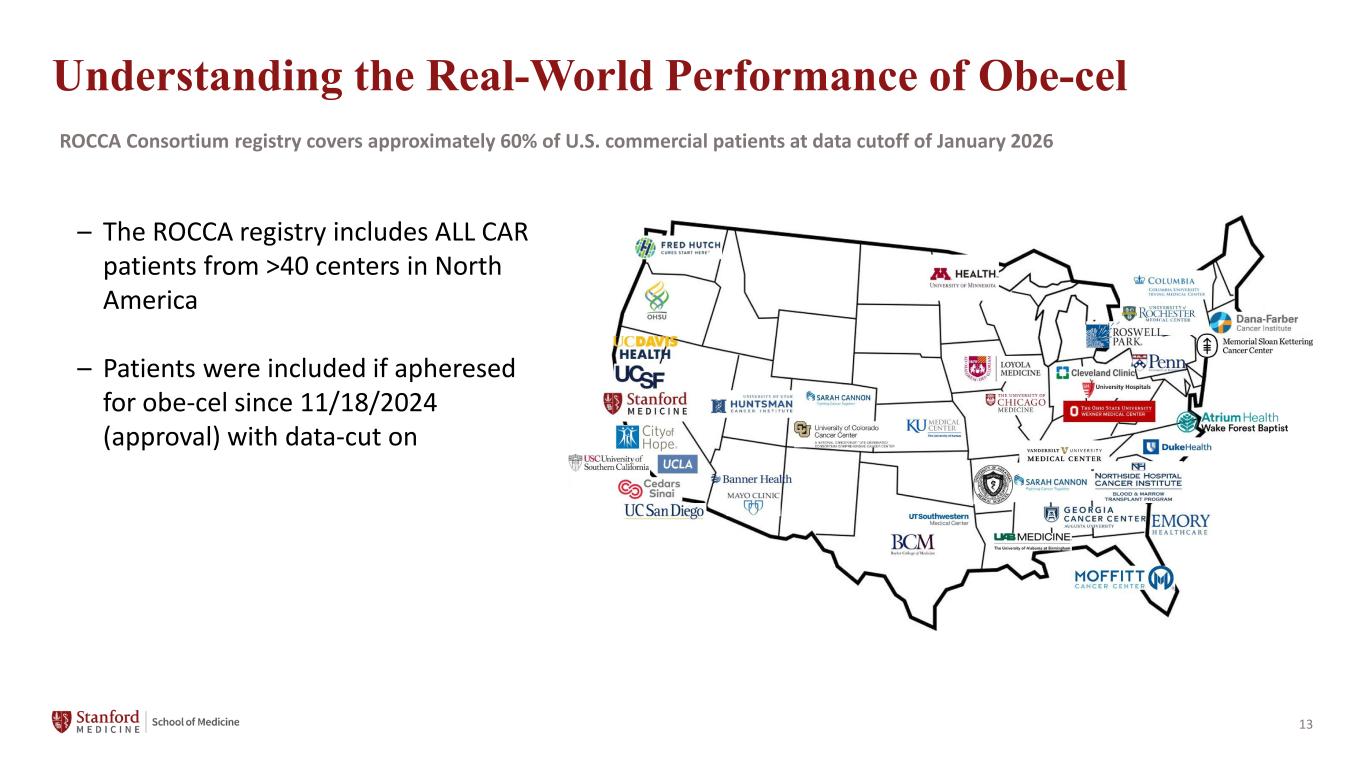
13 – The ROCCA registry includes ALL CAR patients from >40 centers in North America – Patients were included if apheresed for obe-cel since 11/18/2024 (approval) with data-cut on Understanding the Real-World Performance of Obe-cel ROCCA Consortium registry covers approximately 60% of U.S. commercial patients at data cutoff of January 2026
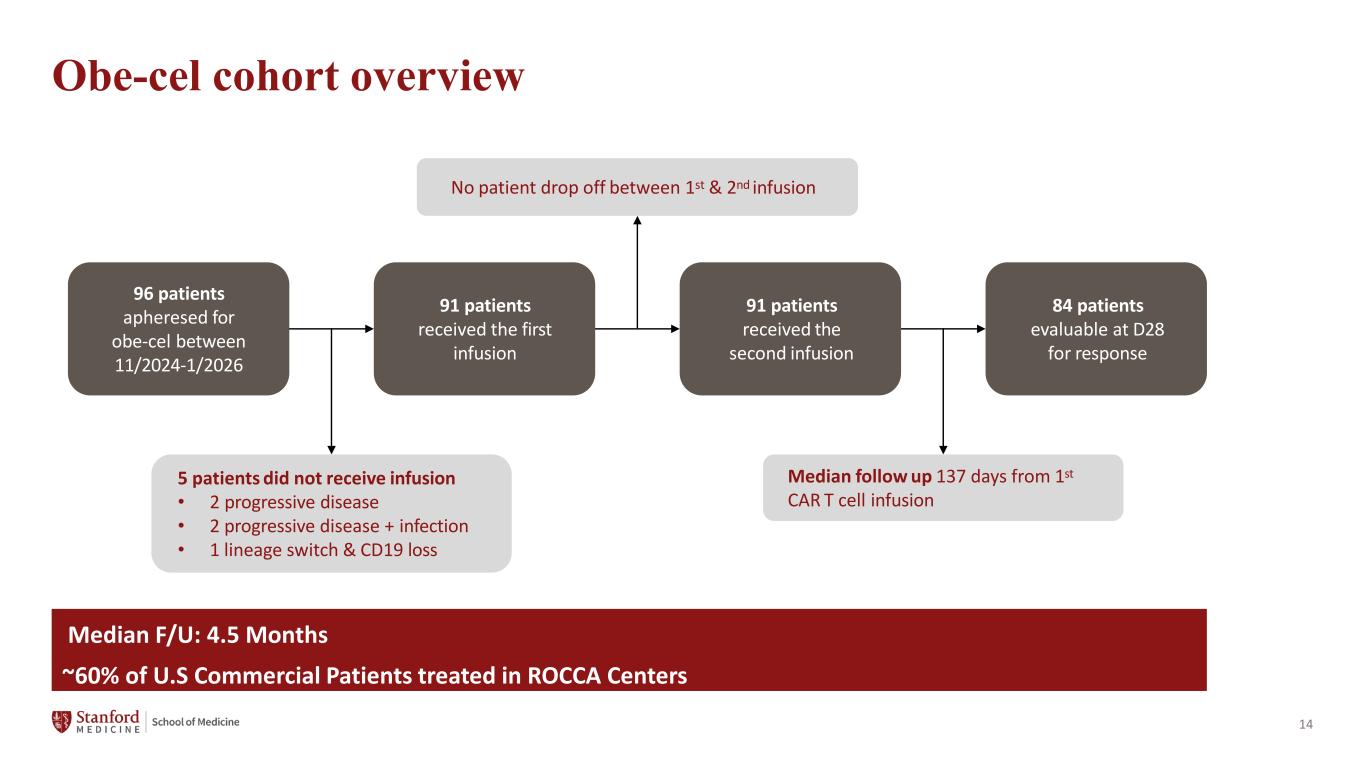
14 Obe-cel cohort overview 96 patients apheresed for obe-cel between 11/2024-1/2026 91 patients received the first infusion 91 patients received the second infusion 84 patients evaluable at D28 for response 5 patients did not receive infusion • 2 progressive disease • 2 progressive disease + infection • 1 lineage switch & CD19 loss No patient drop off between 1st & 2nd infusion Median follow up 137 days from 1st CAR T cell infusion Median F/U: 4.5 Months ~60% of U.S Commercial Patients treated in ROCCA Centers
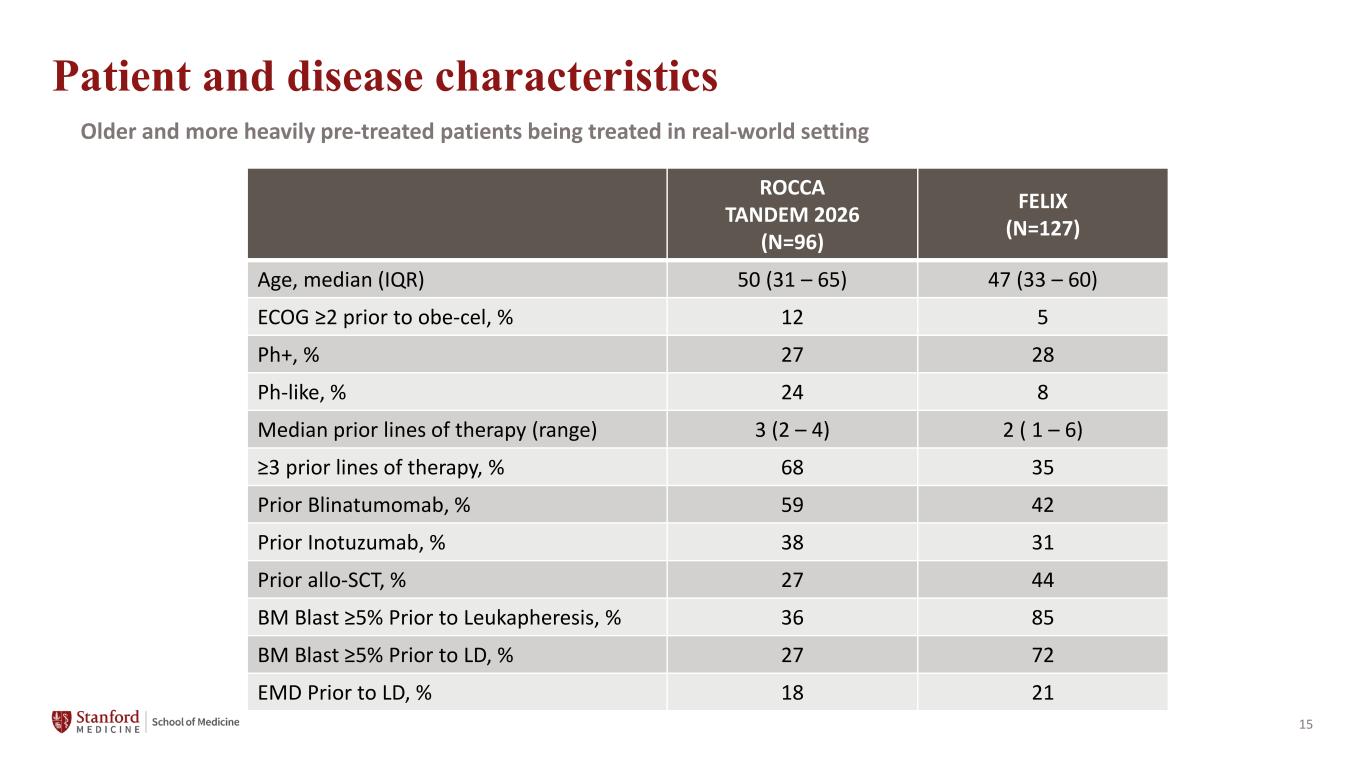
15 Patient and disease characteristics Older and more heavily pre-treated patients being treated in real-world setting ROCCA TANDEM 2026 (N=96) FELIX (N=127) Age, median (IQR) 50 (31 – 65) 47 (33 – 60) ECOG ≥2 prior to obe-cel, % 12 5 Ph+, % 27 28 Ph-like, % 24 8 Median prior lines of therapy (range) 3 (2 – 4) 2 ( 1 – 6) ≥3 prior lines of therapy, % 68 35 Prior Blinatumomab, % 59 42 Prior Inotuzumab, % 38 31 Prior allo-SCT, % 27 44 BM Blast ≥5% Prior to Leukapheresis, % 36 85 BM Blast ≥5% Prior to LD, % 27 72 EMD Prior to LD, % 18 21
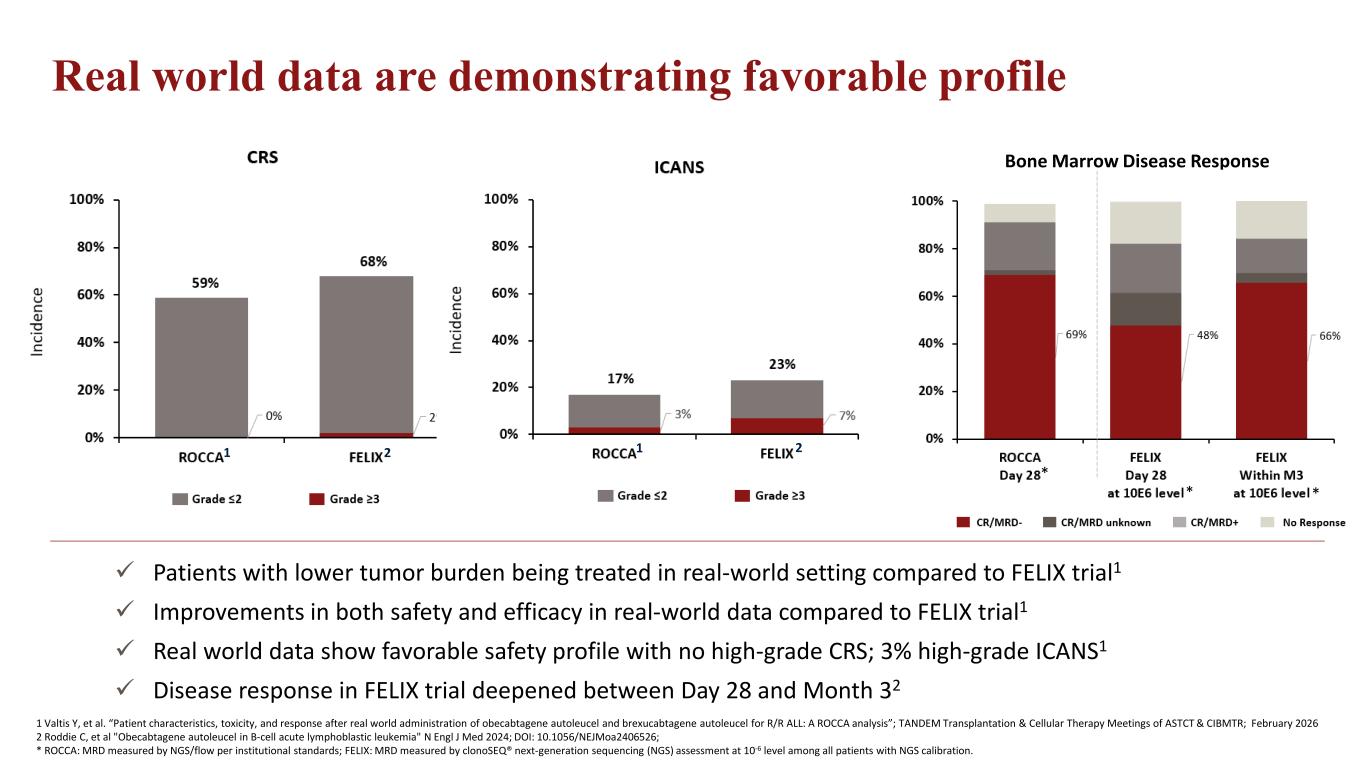
Real world data are demonstrating favorable profile 1 Valtis Y, et al. “Patient characteristics, toxicity, and response after real world administration of obecabtagene autoleucel and brexucabtagene autoleucel for R/R ALL: A ROCCA analysis”; TANDEM Transplantation & Cellular Therapy Meetings of ASTCT & CIBMTR; February 2026 2 Roddie C, et al "Obecabtagene autoleucel in B-cell acute lymphoblastic leukemia" N Engl J Med 2024; DOI: 10.1056/NEJMoa2406526; * ROCCA: MRD measured by NGS/flow per institutional standards; FELIX: MRD measured by clonoSEQ® next-generation sequencing (NGS) assessment at 10-6 level among all patients with NGS calibration. Bone Marrow Disease Response Patients with lower tumor burden being treated in real-world setting compared to FELIX trial1 Improvements in both safety and efficacy in real-world data compared to FELIX trial1 Real world data show favorable safety profile with no high-grade CRS; 3% high-grade ICANS1 Disease response in FELIX trial deepened between Day 28 and Month 32 1 2 1 2 * * *

17 • Obe-cel’s high response rate combined with minimal side effect profile allows us to treat older, frailer patients, and to treat patients exclusively in the outpatient setting. • Obe-cel has been mostly used in patients with a variety of disease burdens, from MRD-negative to high-burden disease, along with patients with both medullary and extra-medullary leukemia. • Our strategy is to consider obe-cel for all patients with relapsed or refractory B-ALL. We typically use bridging therapy for patients with morphologic disease or high levels of MRD prior to apheresis. We do not routinely offer allogeneic transplant following obe-cel. AUCATZYL is redefining CAR T eligibility
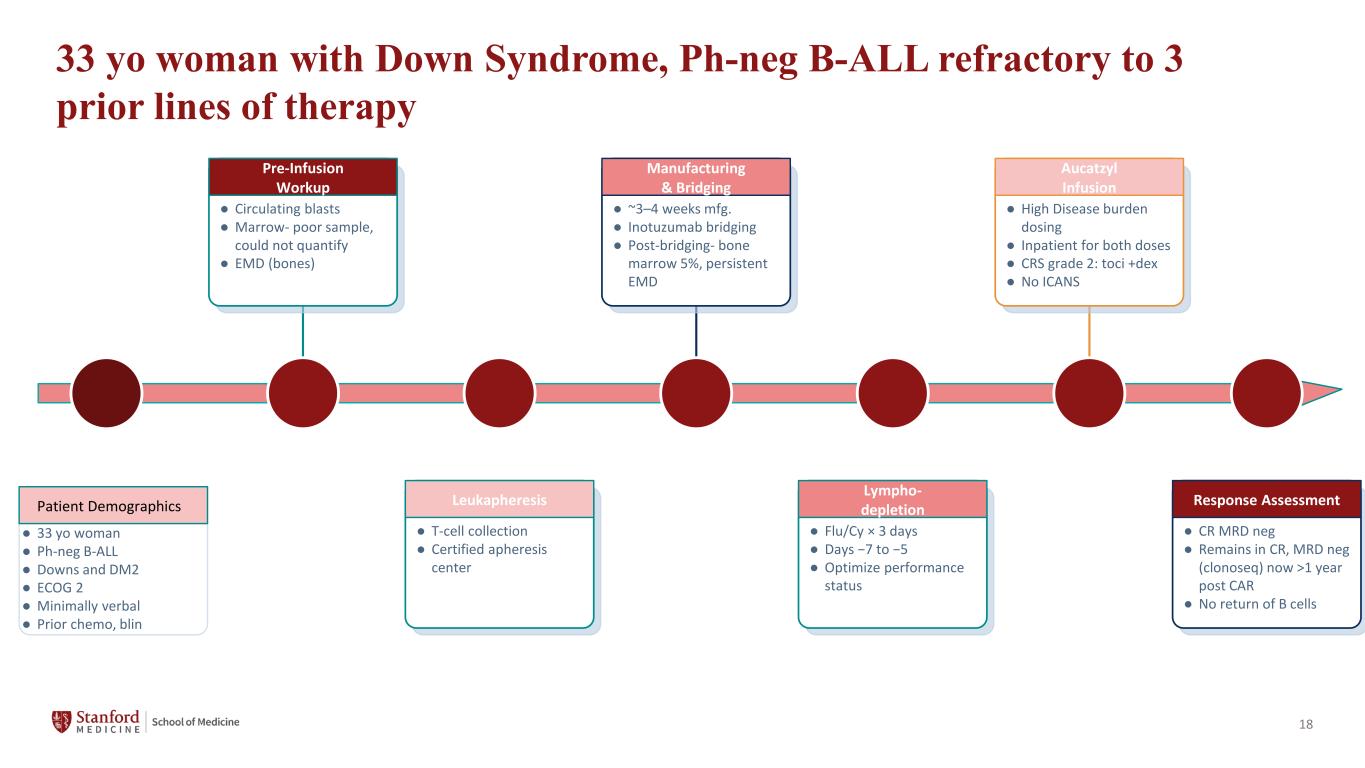
18 33 yo woman with Down Syndrome, Ph-neg B-ALL refractory to 3 prior lines of therapy Patient Demographics ● 33 yo woman ● Ph-neg B-ALL ● Downs and DM2 ● ECOG 2 ● Minimally verbal ● Prior chemo, blin Pre-Infusion Workup ● Circulating blasts ● Marrow- poor sample, could not quantify ● EMD (bones) Leukapheresis ● T-cell collection ● Certified apheresis center Manufacturing & Bridging ● ~3–4 weeks mfg. ● Inotuzumab bridging ● Post-bridging- bone marrow 5%, persistent EMD Lympho- depletion ● Flu/Cy × 3 days ● Days −7 to −5 ● Optimize performance status Aucatzyl Infusion ● High Disease burden dosing ● Inpatient for both doses ● CRS grade 2: toci +dex ● No ICANS Response Assessment ● CR MRD neg ● Remains in CR, MRD neg (clonoseq) now >1 year post CAR ● No return of B cells
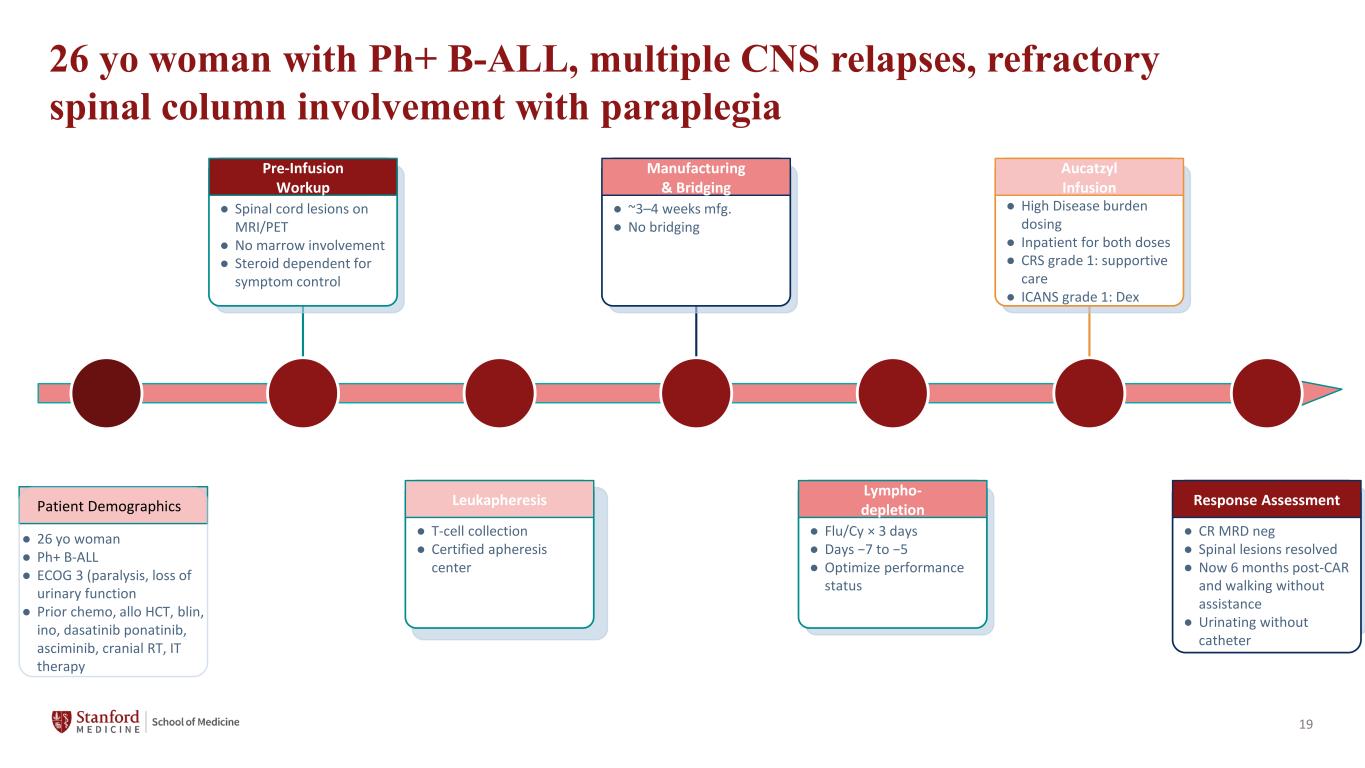
19 26 yo woman with Ph+ B-ALL, multiple CNS relapses, refractory spinal column involvement with paraplegia Patient Demographics Pre-Infusion Workup ● Spinal cord lesions on MRI/PET ● No marrow involvement ● Steroid dependent for symptom control Leukapheresis ● T-cell collection ● Certified apheresis center Manufacturing & Bridging ● ~3–4 weeks mfg. ● No bridging Lympho- depletion ● Flu/Cy × 3 days ● Days −7 to −5 ● Optimize performance status Aucatzyl Infusion ● High Disease burden dosing ● Inpatient for both doses ● CRS grade 1: supportive care ● ICANS grade 1: Dex Response Assessment ● CR MRD neg ● Spinal lesions resolved ● Now 6 months post-CAR and walking without assistance ● Urinating without catheter ● 26 yo woman ● Ph+ B-ALL ● ECOG 3 (paralysis, loss of urinary function ● Prior chemo, allo HCT, blin, ino, dasatinib ponatinib, asciminib, cranial RT, IT therapy

20 ROCCA Consortium Research Domains 1 Efficacy & Survival Outcomes ▸~150-200 obe-cel treated patients ▸Populations of interest: ▸Older adults ▸AYAs ▸CNS/EMD 2 Toxicity: CRS & ICANS Management ▸General toxicity assessment ▸Predictors of toxicity ▸Toxicity management strategies 3 Prognostic Biomarkers ▸Correlative studies: ▸Persistence ▸MRD ▸B cell aplasia ▸T cell phenotypes 4 Bridging Therapy Strategy ▸Optimal bridging ▸Pre vs post-bridging disease assessments 5 Post-CAR Consolidation & Maintenance ▸TKI Maintenance in Ph+ ALL ▸Allo HCT ▸Menin inhibitor maintenance 6 Healthcare Utilization ▸CAR T administration setting ▸Access and equity ▸LTFU/QOL 7 Disease burden Considerations 8 Comparative Analyses ▸Obe-cel superiority over alternative CAR/immunotherapy agents ▸CAR vs allo HCT studies ▸Earlier CAR administration ▸Duration of remission among MRD-negative patients
A U T O L U S I N V E S T O R E V E N T · 2 0 2 6 Obe-cel in Frontline Consolidation for Adult B-ALL The Opportunity, the Evidence, and the Path Forward P R E S E N T I N G F A C U L T Y Elias Jabbour, MD D.B. Lane Cancer Research Fund Distinguished Professor in Leukemia Research Section Chief Acute Lymphoblastic Leukemia MD Anderson Cancer Center Houston, Texas Adult B-ALL Frontline immunotherapy CAR-T consolidation The views expressed represent the independent scientific perspective of Dr. Jabbour. Dr. Jabbour discloses a consulting/advisory relationship with Autolus Therapeutics.

Department of Leukemia Division of Cancer Medicine · Houston, Texas Why Frontline? The Biological and Clinical Rationale for Earlier CAR-T The disease burden argument Low burden Best CAR-T outcomes consistently observed in patients with minimal disease at infusion — established across all approved CD19 products ~70% MRD-negative CR rate achievable with modern induction (hyper-CVAD + InO + blina) — an ideal pre- CAR-T platform T cell fitness Apheresis from patients in early CR1 is less exhausted, less lymphopenic, and yields a higher- quality CAR-T product than R/R patients No escape CD19 expression is uniformly high in newly diagnosed B-ALL — antigen loss risk is dramatically lower than in multiply relapsed disease Jabbour et al., Leukemia & Lymphoma 2025 — published position "CAR T-cell therapies as a consolidation approach have shown positive outcomes. Newer generation CAR T-cell products like obecabtagene autoleucel appear as effective and safer." Senapati, Kantarjian, Jabbour et al., Leukemia & Lymphoma, 2025 The MDACC consolidation framework MDACC has pioneered immunotherapy-based induction (hyper-CVAD/mini-hCVD + InO + blinatumomab) achieving deep CR1 in the majority of adults — creating the ideal disease-burden state for CAR-T consolidation. The MRD-negativity gateway Patients achieving MRD-negative CR1 with modern induction may not need allo-SCT if followed by durable CAR-T consolidation. This is the most significant paradigm shift in adult ALL in decades. The independent investigator view Jabbour's group has directly validated the frontline consolidation hypothesis in peer-reviewed literature and active clinical investigation — the highest-credibility endorsement of this strategy. Senapati J, Kantarjian H, Jabbour E et al. Leukemia & Lymphoma 2025; MDACC frontline consolidation trial

Department of Leukemia Division of Cancer Medicine · Houston, Texas The Frontline B-ALL Landscape Is Being Redefined by Immunotherapy Era 1 · 1980–2010 Intensive chemo ± allo-SCT Hyper-CVAD dominant ~40% OS at 5 years › Era 2 · 2011–2020 TKI + chemo (Ph+ ALL) Ponatinib combinations improved Ph+ outcomes › Era 3 · 2021–2024 Immunotherapy in consolidation Blinatumomab approved FL Inotuzumab combinations › Era 4 · 2025+ CAR-T frontline consolidation SCT-sparing intent for high-risk & MRD+ patients What changed the calculus for frontline CAR-T 01 Modern induction achieves MRD-negative CR1 in ~70% of patients — the optimal pre-CAR-T state, achievable in real practice today 02 R/R data prove disease burden at infusion is the single strongest predictor of durable response — frontline is the optimal intervention window 03 Obe-cel's low Grade 3+ CRS/ICANS profile makes frontline administration feasible where competitor toxicity profiles would preclude it 04 MRD-guided intensification is rapidly becoming the NCCN/ASH framework — CAR-T consolidation maps directly onto that clinical decision point Senapati et al., Leukemia & Lymphoma 2025; Jabbour et al., JCO 2024; NCCN B-ALL Guidelines v2.2025
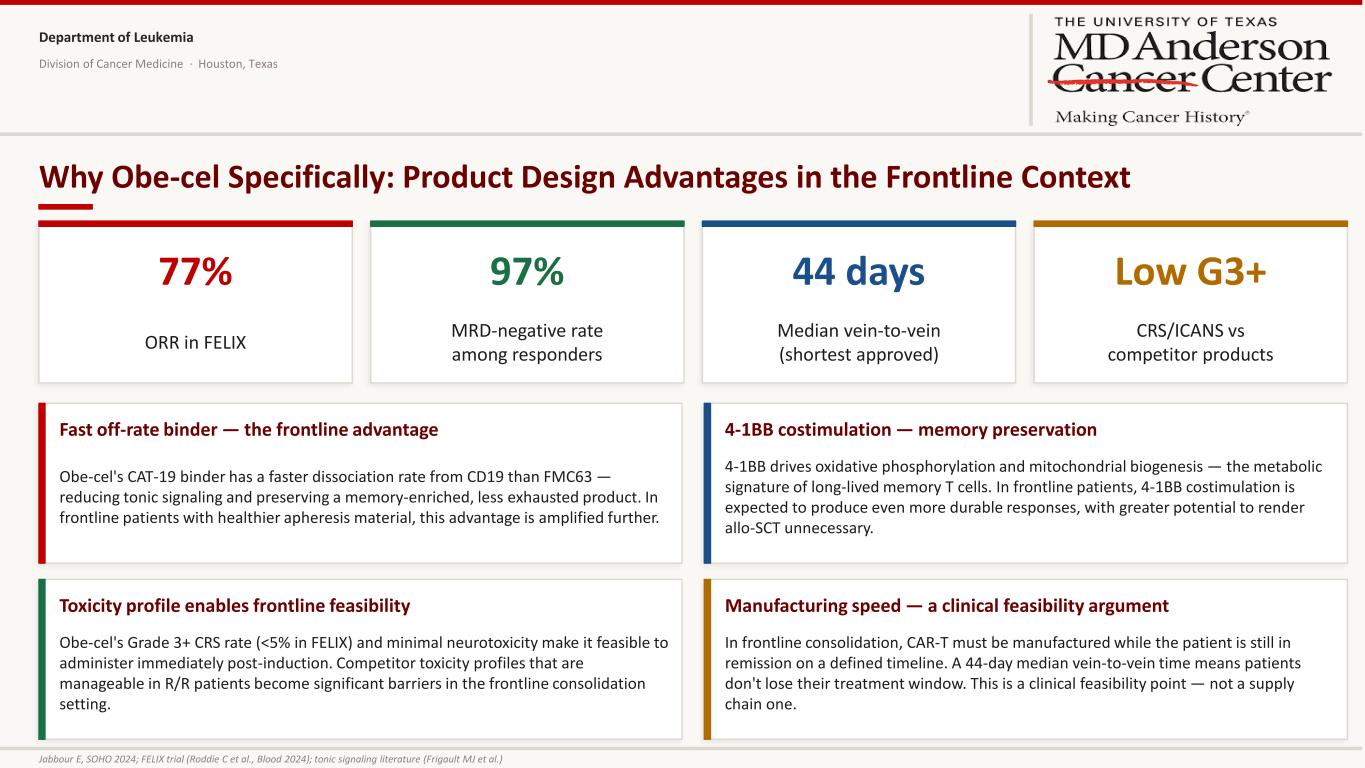
Department of Leukemia Division of Cancer Medicine · Houston, Texas Why Obe-cel Specifically: Product Design Advantages in the Frontline Context 77% ORR in FELIX 97% MRD-negative rate among responders 44 days Median vein-to-vein (shortest approved) Low G3+ CRS/ICANS vs competitor products Fast off-rate binder — the frontline advantage Obe-cel's CAT-19 binder has a faster dissociation rate from CD19 than FMC63 — reducing tonic signaling and preserving a memory-enriched, less exhausted product. In frontline patients with healthier apheresis material, this advantage is amplified further. 4-1BB costimulation — memory preservation 4-1BB drives oxidative phosphorylation and mitochondrial biogenesis — the metabolic signature of long-lived memory T cells. In frontline patients, 4-1BB costimulation is expected to produce even more durable responses, with greater potential to render allo-SCT unnecessary. Toxicity profile enables frontline feasibility Obe-cel's Grade 3+ CRS rate (<5% in FELIX) and minimal neurotoxicity make it feasible to administer immediately post-induction. Competitor toxicity profiles that are manageable in R/R patients become significant barriers in the frontline consolidation setting. Manufacturing speed — a clinical feasibility argument In frontline consolidation, CAR-T must be manufactured while the patient is still in remission on a defined timeline. A 44-day median vein-to-vein time means patients don't lose their treatment window. This is a clinical feasibility point — not a supply chain one. Jabbour E, SOHO 2024; FELIX trial (Roddie C et al., Blood 2024); tonic signaling literature (Frigault MJ et al.)

Department of Leukemia Division of Cancer Medicine · Houston, Texas Patient Selection Framework: Who Benefits Most from Frontline Consolidation Priority patient populations HIGHEST PRIORITYAdverse genomics in CR1 CRLF2r, KMT2Ar, TP53 mutation, complex karyotype, hypodiploidy — these patients have poor outcomes with SCT alone. CAR-T offers a novel mechanism independent of cytogenetic risk. HIGHEST PRIORITYMRD-positive after induction Failure to achieve MRD negativity after cycle 2 signals dramatically worse prognosis. CAR-T consolidation before overt relapse is the optimal intervention point. HIGH PRIORITYTransplant-ineligible adults in CR1 Older adults and those with comorbidities or no matched donor in CR1 currently have no durable consolidation option. Obe-cel's safety profile makes it the leading candidate. EMERGINGSCT-eligible patients preferring avoidance A growing proportion of adults in CR1 prefer to avoid transplant morbidity. Shared decision- making increasingly incorporates CAR-T as a consolidation option. MDACC proposed treatment schema (Jabbour framework) 1 Induction Hyper-CVAD / mini-hCVD + Inotuzumab + Blinatumomab 2 MRD assessment at C2 NGS-MRD: risk-stratify for consolidation pathway selection 3 Leukapheresis in CR1 Collect at remission — optimal T cell fitness window 4 Obe-cel consolidation CAR-T infusion ~44 days post-collection Post-lymphodepleting chemotherapy 5 Response-adapted follow-up MRD-neg CR → surveillance MRD-persistent → escalation or SCT Senapati et al., Leukemia & Lymphoma 2025; MDACC Phase 2 frontline consolidation trial; Jabbour EJ, frontline ALL review 2024
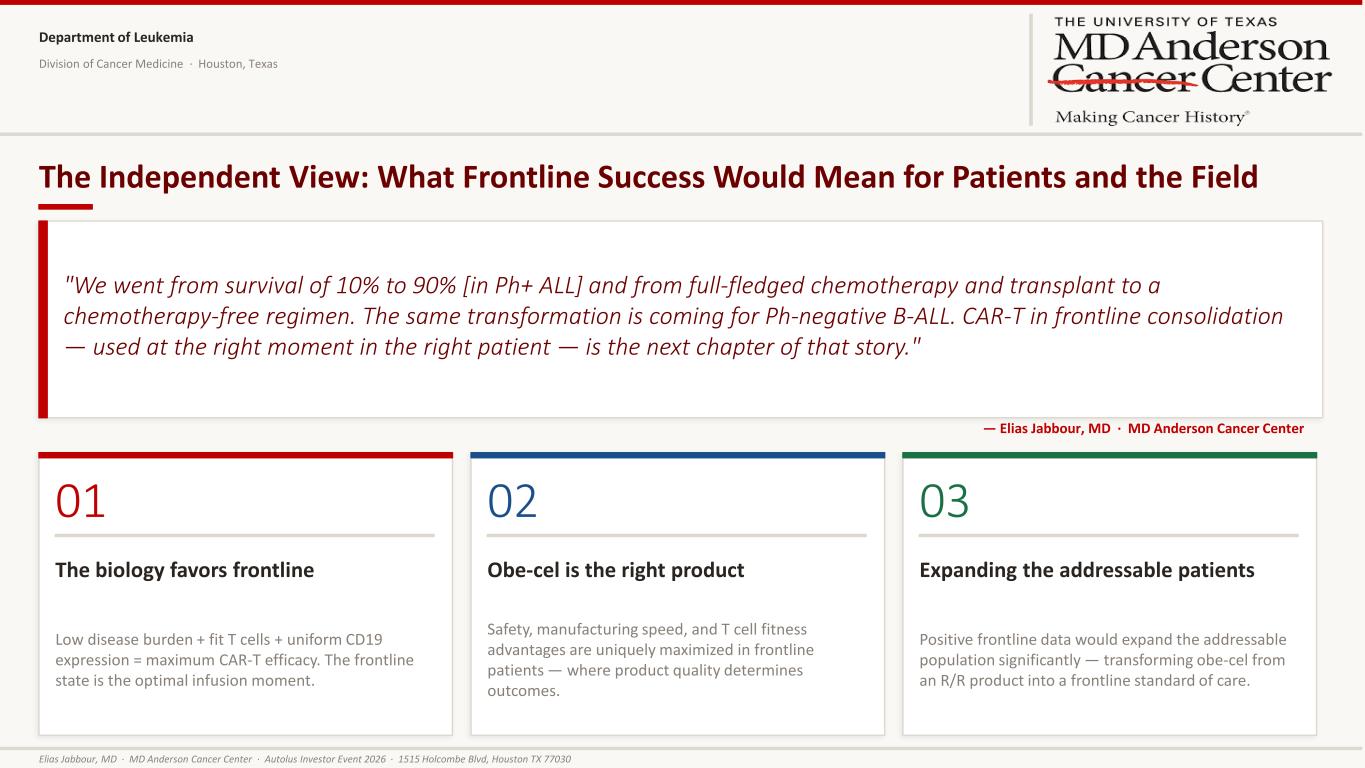
Department of Leukemia Division of Cancer Medicine · Houston, Texas The Independent View: What Frontline Success Would Mean for Patients and the Field "We went from survival of 10% to 90% [in Ph+ ALL] and from full-fledged chemotherapy and transplant to a chemotherapy-free regimen. The same transformation is coming for Ph-negative B-ALL. CAR-T in frontline consolidation — used at the right moment in the right patient — is the next chapter of that story." — Elias Jabbour, MD · MD Anderson Cancer Center 01 The biology favors frontline Low disease burden + fit T cells + uniform CD19 expression = maximum CAR-T efficacy. The frontline state is the optimal infusion moment. 02 Obe-cel is the right product Safety, manufacturing speed, and T cell fitness advantages are uniquely maximized in frontline patients — where product quality determines outcomes. 03 Expanding the addressable patients Positive frontline data would expand the addressable population significantly — transforming obe-cel from an R/R product into a frontline standard of care. Elias Jabbour, MD · MD Anderson Cancer Center · Autolus Investor Event 2026 · 1515 Holcombe Blvd, Houston TX 77030

Use and Research Priorities of CAR T-Cells for Pediatric/AYA ALL; Catulus Data to Date/Plans Michael A. Pulsipher, MD Division Chief, Pediatric Hematology and Oncology Director, Children’s and Adolescent Cancer Initiative at Huntsman Cancer Institute and Intermountain Primary Children’s Hospital IPCH-HCI Presidential Chair in Pediatric Oncology and Hematology

Overview • Critical Role for CD19 targeted CAR T-cells in Relapsed/ Refractory ALL for Children, Adolescents and Young Adults (CAYA) • Research Gaps and Challenges for CAR T-cell use in CAYA • Results to date of Catulus Phase Ib (Auto1-PY1) • Plans moving forward for extension and regulatory approval: • AALL2523/AFFI2522: A Single-Arm, Open-Label, Multi-Centre, Phase 1b/2 Study Evaluating the Safety and Preliminary Efficacy of AUTO1 (Obe-cel) in Pediatric Patients with CD19-Positive Relapsed/Refractory (r/r) B cell Acute Lymphoblastic Leukemia (B ALL) and Aggressive Mature B cell NON-HODGKIN Lymphoma (B NHL)

Current Role of CAR T-cells in CAYA with B-ALL • FDA Approved Tisa-cel in 2017 for relapsed/refractory B-ALL • Originally use mainly for rescue post-HCT or after multiple remissions • Better outcomes when used with low disease burden/earlier in the Rx course • Increased in usage in the use each year 2018-2022 • Use shifting more to primary refractory or secondary refractory • Role of HCT after 4-IBB CAR T-cells much debated • Loss of B-cell aplasia and NGS-MRD assays sometimes used to guide therapy • Survival has improved significantly! • Previous survival <10% • Now, 5-year EFS approximately 40%, OS exceeds 60%

Research Questions and Needs in CAYA • Early first relapse of ALL has dismal outcomes • Would planned use of CAR T-cells at first relapse decrease toxicity/improve survival? • MRD level disease post-consolidation generally requires transplant • Can use of CART cells in this patient population avoid HSCT and improve outcomes? • AALL 1721 outcomes pending • Special populations need CAR T-cell therapy early • Children/AYA with Down Syndrome • Frail children—history of multiple complications with chemotherapy, organ damage, infections, etc. • Genetic subtypes susceptible to secondary malignancies with chemotherapy/TBI approaches (hypodiploid ALL, TP53 germline mutations) • Infants with VHR B-ALL—generally very poor prognosis

Treatment of paediatric patients with relapsed/refractory B-cell acute lymphoblastic leukemia (R/R B-ALL) with obecabtagene autoleucel (obe-cel), a CD19-directed chimeric antigen receptor (CAR) T-cell therapy: preliminary findings from the Phase Ib CATULUS trial Sara Ghorashian1*, Michael A Pulsipher2, Juliana Silva1, Alba Rubio-San-Simón3, Amanda Lipsitt4, Denise Bonney5, Cristina Díaz-de-Heredia6, Geoff Shenton7, Justin Shang8, Kapil Saxena8, Wolfram Brugger9, Pierre Lao-Sirieix10, Shannon L Maude11 1. Great Ormond Street Hospital for Children NHS Foundation Trust, London, UK; 2. Intermountain Primary Children’s Hospital, Huntsman Cancer Institute, Spencer Fox Eccles School of Medicine at the University of Utah, Salt Lake City, UT, USA; 3. Hospital Universitario Niño Jesús, Madrid, Spain; 4. Methodist Children’s Hospital, San Antonio, TX, USA; 5. Royal Manchester Children’s Hospital, Paediatric Oncology and Haematology Research Team, St Mary’s Hospital, Manchester, UK; 6. Pediatric Hematology and Oncology Department, Hospital Vall d’Hebron and Vall d’Hebron Research Institute, Barcelona, Spain; 7. Great North Children’s Hospital, Paediatric Oncology Clinical Trials Team, Newcastle Upon Tyne, UK; 8. Autolus Therapeutics, Rockville, MD, USA; 9. Autolus Therapeutics, Munich, Germany; 10. Autolus Therapeutics, London, UK; 11. Children’s Hospital of Philadelphia, Philadelphia, PA, USA. American Society of Hematology (ASH) Annual Meeting 2025: This presentation has been adapted from Poster 3337 originally presented at the conference Acknowledgments This study was sponsored by Autolus Therapeutics PLC. Third-party medical writing assistance, under the direction of authors, was provided by Emily Fang, MPharm, of Ashfield MedComms (Manchester, UK), an Inizio company, and was funded by Autolus Therapeutics PLC. *Presenter. This presentation is adapted from Poster 3337, originally presented at the American Society of Hematology (ASH) Annual Meeting 2025, December 6-9 in Orlando, FL, USA.
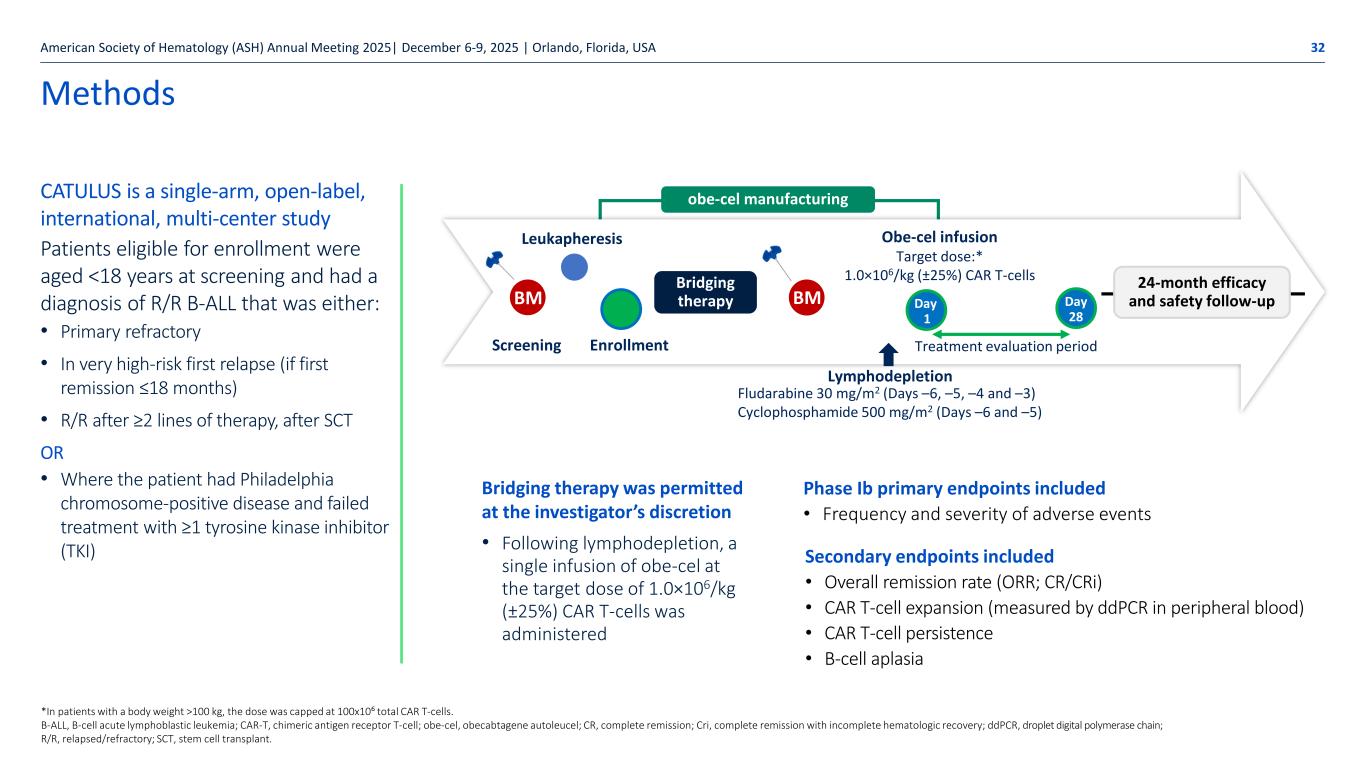
Autolus.com Leukapheresis Obe-cel infusion Target dose:* 1.0×106/kg (±25%) CAR T-cells Day 1 Day 28 Fludarabine 30 mg/m2 (Days –6, –5, –4 and –3) Cyclophosphamide 500 mg/m2 (Days –6 and –5) obe-cel manufacturing BMBM Bridging therapy Screening Enrollment Lymphodepletion Treatment evaluation period 24-month efficacy and safety follow-up Methods CATULUS is a single-arm, open-label, international, multi-center study Patients eligible for enrollment were aged <18 years at screening and had a diagnosis of R/R B-ALL that was either: • Primary refractory • In very high-risk first relapse (if first remission ≤18 months) • R/R after ≥2 lines of therapy, after SCT OR • Where the patient had Philadelphia chromosome-positive disease and failed treatment with ≥1 tyrosine kinase inhibitor (TKI) American Society of Hematology (ASH) Annual Meeting 2025| December 6-9, 2025 | Orlando, Florida, USA 32 Bridging therapy was permitted at the investigator’s discretion • Following lymphodepletion, a single infusion of obe-cel at the target dose of 1.0×106/kg (±25%) CAR T-cells was administered *In patients with a body weight >100 kg, the dose was capped at 100x10⁶ total CAR T-cells. B-ALL, B-cell acute lymphoblastic leukemia; CAR-T, chimeric antigen receptor T-cell; obe-cel, obecabtagene autoleucel; CR, complete remission; Cri, complete remission with incomplete hematologic recovery; ddPCR, droplet digital polymerase chain; R/R, relapsed/refractory; SCT, stem cell transplant. Secondary endpoints included • Overall remission rate (ORR; CR/CRi) • CAR T-cell expansion (measured by ddPCR in peripheral blood) • CAR T-cell persistence • B-cell aplasia Phase Ib primary endpoints included • Frequency and severity of adverse events

Autolus.com Baseline Characteristics As of August 6, 2025 (median follow-up: 8.8 months; range: 0.5–18.4), 23/25 enrolled patients (82.1%) with R/R B-ALL were infused with obe-cel One patient was not infused per physician decision, and one patient was pending obe-cel infusion at the time of data cut-off • All 23 patients received obe-cel at the target dose; all products were in specification, with no manufacturing failures – Median time from leukapheresis to product certification was 19 days (range: 15–31) • Overall, all 23 patients received bridging therapy with: – chemotherapy (n=18), – chemotherapy and inotuzumab ozogamicin (n=3) – chemotherapy and a tyrosine kinase inhibitor (n=2) American Society of Hematology (ASH) Annual Meeting 2025| December 6-9, 2025 | Orlando, Florida, USA 33 Demographics and baseline characteristics at screening All infused Patients, B-ALL Cohort (N=23) Age in years, median (range) 9.2 (0.8–17.8) <2, n (%) 2 (8.7) ≥2–<6, n (%) 6 (26.1) ≥6–<12, n (%) 5 (21.7) ≥12, n (%) 10 (43.5) Male/female, n (%) 11/12 (47.8/52.2) Ethnicity, n (%) Hispanic or Latino 6 (26.1) Not Hispanic or Latino 17 (73.9) Down syndrome, n (%) 1 (4.3) KMT2A rearrangement, n (%) 2 (8.7) Philadelphia chromosome-positive, n (%) 3 (13.0) Prior treatment, n (%) Prior allogeneic SCT 3 (13.0) Prior blinatumomab 6 (26.1) Prior inotuzumab ozogamicin 0 Primary refractory disease, n (%) 6 (26.1) First relapse, n (%) 5 (21.7) First relapse in BM, <36 months after initial diagnosis 2 (8.7) First relapse isolated EMD, <18 months after initial diagnosis 2 (8.7) Other 1 (4.3) Second or greater relapse, n (%) 12 (52.2) BM blast % at screening, median (range) 5.0 (0–95) ≥5% BM blast % at screening, n (%) 13 (56.5) <5% BM blast % at screening,* n (%) 10 (43.5) CR/CRi‡ at screening, n (%) 5 (21.7) MRD-positive (≥0.01%–<1% leukemic cells, per local assessment) 5 (21.7) MRD-negative (<0.01%) 0 Not CR/CRi‡ at screening, n (%) 18 (78.3) BM and/or blood disease with/without EMD 14 (60.9) Isolated EMD 4 (17.4) EMD disease at screening, n (%) 6 (26.1) Central nervous system 5 (21.7) Testis 2 (8.7) Other 1 (4.3) *Four patients had isolated EMD and one patient had MRD ≥1% by molecular testing. ‡Morphologic CR/CRi, defined as <5% BM blasts or <1% by molecular testing and no EMD. B-ALL, B-cell acute lymphoblastic leukemia; BM, bone marrow; CR, complete remission; CRi, complete remission with incomplete hematologic recovery; EMD, extramedullary; MRD, measurable residual disease; R/R, relapsed/refractory; SCT, stem cell transplant.
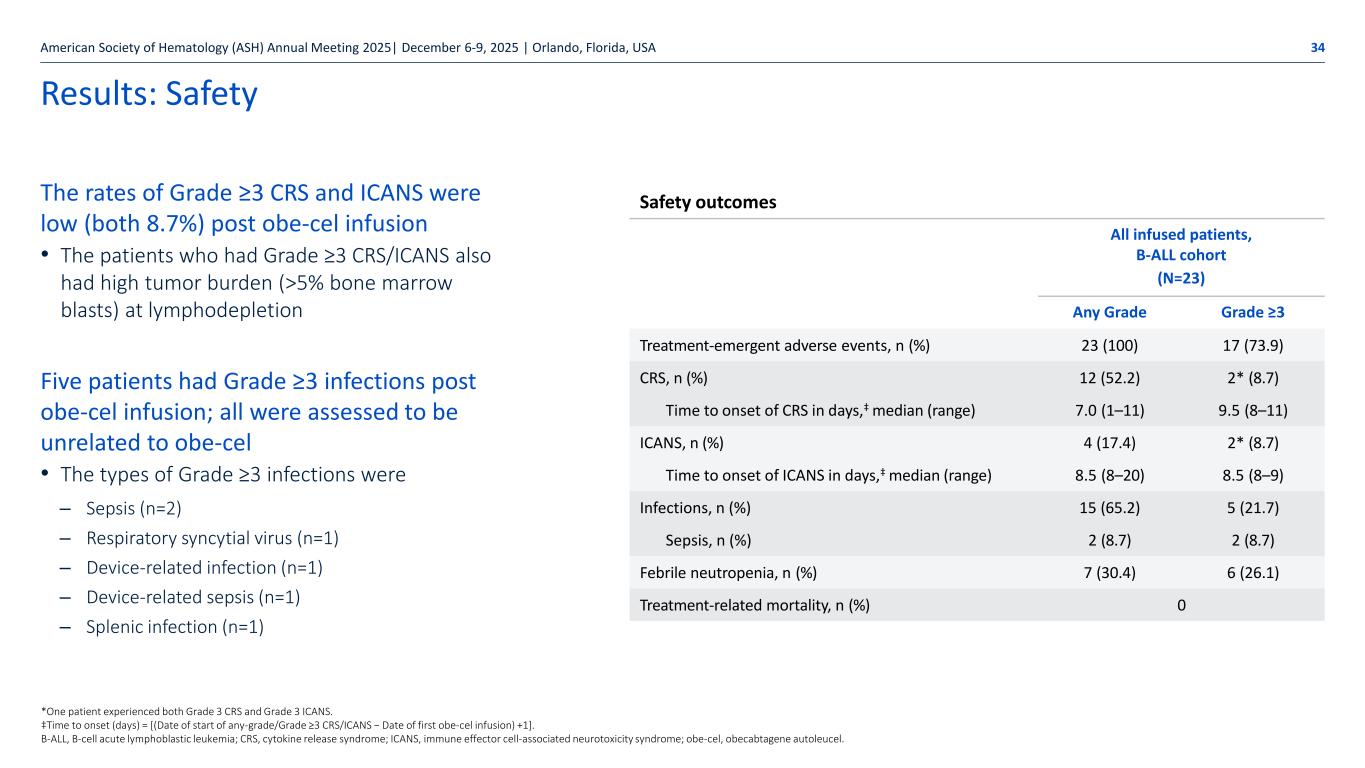
Autolus.com Results: Safety The rates of Grade ≥3 CRS and ICANS were low (both 8.7%) post obe-cel infusion • The patients who had Grade ≥3 CRS/ICANS also had high tumor burden (>5% bone marrow blasts) at lymphodepletion Five patients had Grade ≥3 infections post obe-cel infusion; all were assessed to be unrelated to obe-cel • The types of Grade ≥3 infections were – Sepsis (n=2) – Respiratory syncytial virus (n=1) – Device-related infection (n=1) – Device-related sepsis (n=1) – Splenic infection (n=1) American Society of Hematology (ASH) Annual Meeting 2025| December 6-9, 2025 | Orlando, Florida, USA 34 Safety outcomes All infused patients, B-ALL cohort (N=23) Any Grade Grade ≥3 Treatment-emergent adverse events, n (%) 23 (100) 17 (73.9) CRS, n (%) 12 (52.2) 2* (8.7) Time to onset of CRS in days,‡ median (range) 7.0 (1–11) 9.5 (8–11) ICANS, n (%) 4 (17.4) 2* (8.7) Time to onset of ICANS in days,‡ median (range) 8.5 (8–20) 8.5 (8–9) Infections, n (%) 15 (65.2) 5 (21.7) Sepsis, n (%) 2 (8.7) 2 (8.7) Febrile neutropenia, n (%) 7 (30.4) 6 (26.1) Treatment-related mortality, n (%) 0 *One patient experienced both Grade 3 CRS and Grade 3 ICANS. ‡Time to onset (days) = [(Date of start of any-grade/Grade ≥3 CRS/ICANS − Date of first obe-cel infusion) +1]. B-ALL, B-cell acute lymphoblastic leukemia; CRS, cytokine release syndrome; ICANS, immune effector cell-associated neurotoxicity syndrome; obe-cel, obecabtagene autoleucel.
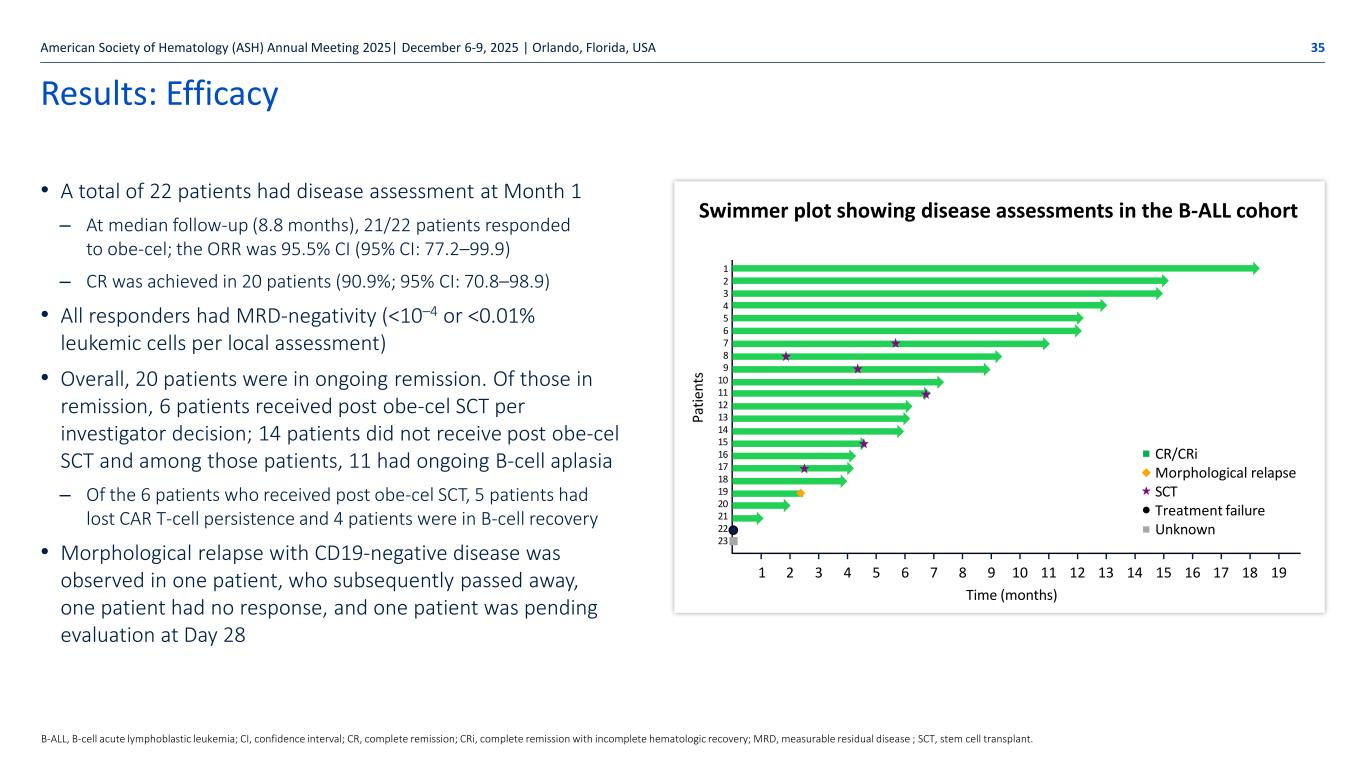
Autolus.com Results: Efficacy • A total of 22 patients had disease assessment at Month 1 – At median follow-up (8.8 months), 21/22 patients responded to obe-cel; the ORR was 95.5% CI (95% CI: 77.2–99.9) – CR was achieved in 20 patients (90.9%; 95% CI: 70.8–98.9) • All responders had MRD-negativity (<10–4 or <0.01% leukemic cells per local assessment) • Overall, 20 patients were in ongoing remission. Of those in remission, 6 patients received post obe-cel SCT per investigator decision; 14 patients did not receive post obe-cel SCT and among those patients, 11 had ongoing B-cell aplasia – Of the 6 patients who received post obe-cel SCT, 5 patients had lost CAR T-cell persistence and 4 patients were in B-cell recovery • Morphological relapse with CD19-negative disease was observed in one patient, who subsequently passed away, one patient had no response, and one patient was pending evaluation at Day 28 American Society of Hematology (ASH) Annual Meeting 2025| December 6-9, 2025 | Orlando, Florida, USA 35 B-ALL, B-cell acute lymphoblastic leukemia; CI, confidence interval; CR, complete remission; CRi, complete remission with incomplete hematologic recovery; MRD, measurable residual disease ; SCT, stem cell transplant. 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 CR/CRi Morphological relapse SCT Treatment failure Unknown Pa tie nt s 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 Time (months) Swimmer plot showing disease assessments in the B-ALL cohort
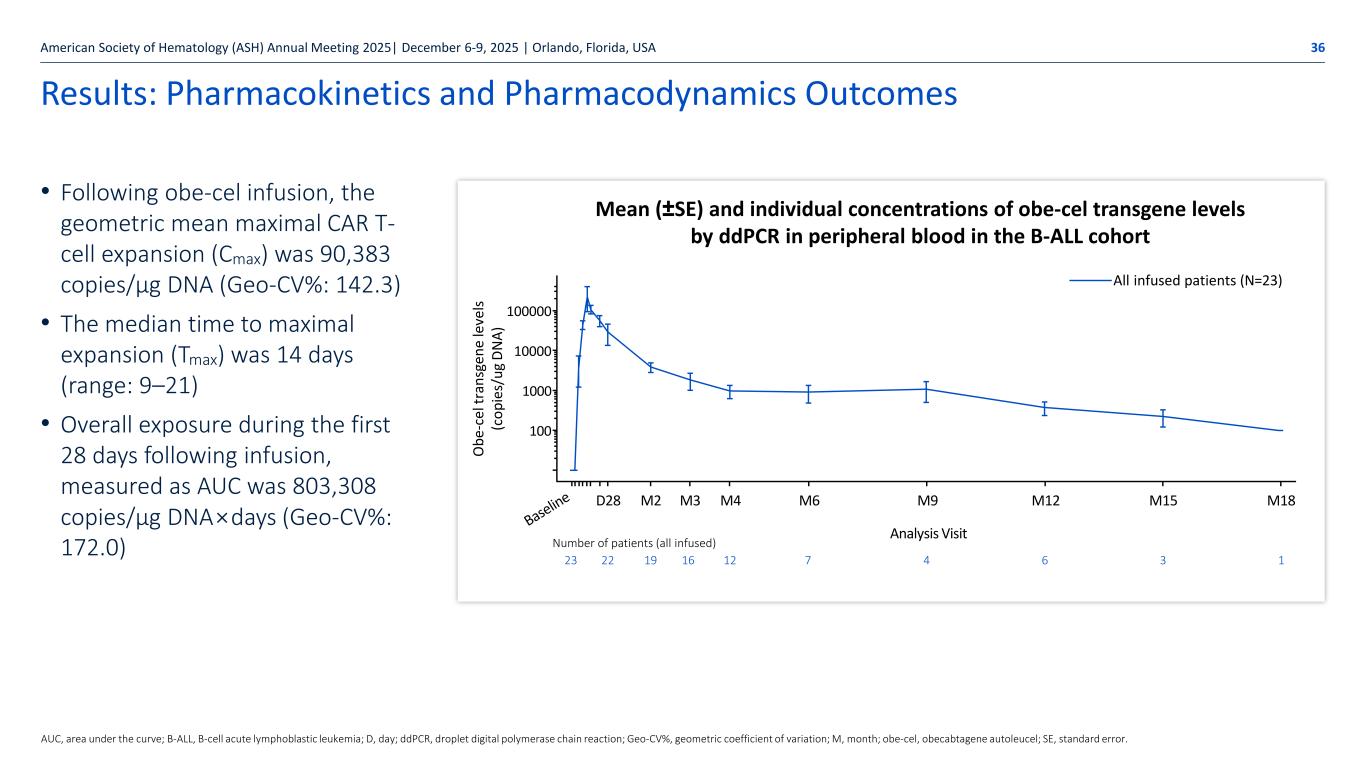
Autolus.com Results: Pharmacokinetics and Pharmacodynamics Outcomes • Following obe-cel infusion, the geometric mean maximal CAR T- cell expansion (Cmax) was 90,383 copies/μg DNA (Geo-CV%: 142.3) • The median time to maximal expansion (Tmax) was 14 days (range: 9–21) • Overall exposure during the first 28 days following infusion, measured as AUC was 803,308 copies/µg DNA×days (Geo-CV%: 172.0) American Society of Hematology (ASH) Annual Meeting 2025| December 6-9, 2025 | Orlando, Florida, USA 36 AUC, area under the curve; B-ALL, B-cell acute lymphoblastic leukemia; D, day; ddPCR, droplet digital polymerase chain reaction; Geo-CV%, geometric coefficient of variation; M, month; obe-cel, obecabtagene autoleucel; SE, standard error. Mean (±SE) and individual concentrations of obe-cel transgene levels by ddPCR in peripheral blood in the B-ALL cohort All infused patients (N=23) M6 M9 M12 M15 M18 O be -c el tr an sg en e le ve ls (c op ie s/ ug DN A) M4M3M2D28 Analysis Visit 100 100000 10000 1000 Number of patients (all infused) 23 22 19 16 12 7 4 6 3 1

Autolus.com Conclusions American Society of Hematology (ASH) Annual Meeting 2025| December 6-9, 2025 | Orlando, Florida, USA • The safety profile of obe-cel in Paediatric patients was consistent with that previously reported in adults, with low rates of high-grade CRS and ICANS • The ORR was high at 95.5% (n=21) and 20 patients were in ongoing remission at data cut-off (median follow-up: 8.8 months) • Manufacture of obe-cel was successful for all patients. Obe-cel effectively expanded (geometric mean: 90,383 copies/μg DNA) after a single infusion at the dose of 1.0×106/kg CAR T-cells • While longer follow-up is needed, results demonstrate promising safety and preliminary efficacy in pediatric patients with R/R B-ALL treated with obe-cel • These preliminary findings support further exploration of obe-cel in pediatric R/R B-ALL. Planning for the Phase II expansion is underway B-ALL, B-cell acute lymphoblastic leukemia; CAR, chimeric antigen receptor;; CRS, cytokine release syndrome; ICANS, immune effector cell-associated neurotoxicity syndrome; obe-cel, obecabtagene autoleucel; ORR, Overall remission rate; R/R, relapsed/refractory.

AALL2523/AFFI2522: A Single-Arm, Open-Label, Multi-Centre, Phase 1b/2 Study Evaluating the Safety and Preliminary Efficacy of AUTO1 (Obe-cel) in Pediatric Patients with CD19-Positive Relapsed/Refractory (r/r) B cell Acute Lymphoblastic Leukemia (B ALL) and Aggressive Mature B cell NON-HODGKIN Lymphoma (B NHL). Study Co-Chairs: Deepa Bhojwani Shannon Maude

• Critical need in first relapse of B-ALL • AALL1331 showed improved outcomes with addition of blinatumomab but • 4% TRM • 40% Grade 3-5 infection • 75% MRD+ • ~40% of patients did not proceed to blina randomization • Intent-to-treat analysis for HR relapse: 2-year EFS 25% • No current indication for CAR T-cell therapy in first relapse Background

• Collaborating with Autolus on their Phase II expansion • Initially focused on high-risk first relapse trial • First collaborating on Ph 2 expansion to support sBLA submission for pediatric indication in r/r B-ALL • Additional 30 ALL patients (for total of 54), with at least 15 slots for high-risk first relapse • Primary Endpoint: • Proportion of patients in remission (CR) per IRRC assessment within 3 months of obe-cel infusion • Secondary endpoints include MRD-negative CR, DOR, EFS, OS, proportion consolidating with SCT, persistence AUTO-PY1 Planned Expansion
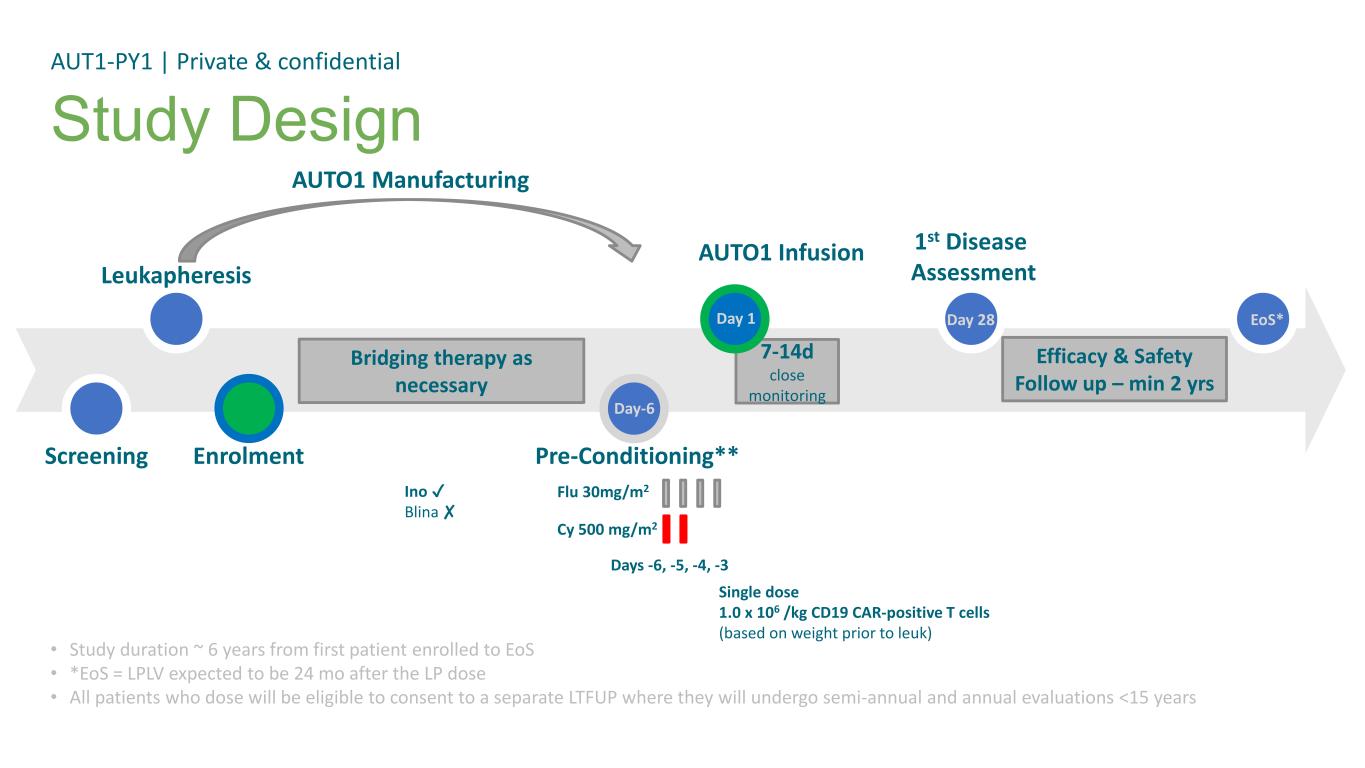
• Study duration ~ 6 years from first patient enrolled to EoS • *EoS = LPLV expected to be 24 mo after the LP dose • All patients who dose will be eligible to consent to a separate LTFUP where they will undergo semi-annual and annual evaluations <15 years Study Design AUT1-PY1 | Private & confidential Cy 500 mg/m2 Screening Leukapheresis Pre-Conditioning** AUTO1 Infusion 1st Disease Assessment Day-6 Day 28 Flu 30mg/m2 Days -6, -5, -4, -3 Enrolment Bridging therapy as necessary AUTO1 Manufacturing Efficacy & Safety Follow up – min 2 yrs EoS* Ino Blina ✘ Single dose 1.0 x 106 /kg CD19 CAR-positive T cells (based on weight prior to leuk) 7-14d close monitoring Day 1

B ALL: r/r CD19-positive B ALL defined as 1 of the following: i. Primary refractory a) NCI high-risk B ALL refractory after first-line induction, consolidation and blinatumomab b) Refractory B ALL if BM disease ≥ 25% after first-line induction and consolidation. c) KMT2A-rearranged infant ALL refractory after first-line induction and blinatumomab ii. First relapse a) COG high-risk first relapse defined as relapse in BM < 36 months after initial diagnosis or an isolated extramedullary relapse < 18 months after initial diagnosis. b) Patients with Down syndrome and infants with KMT2A-r are eligible with first relapse at any time. c) Refractory disease after re-induction for first relapse (+/- blinatumomab) iii. Second or greater relapse iv. Relapsed or refractory post-SCT v. Ph+ ALL: intolerant to or has failed at least 1 TKI or if TKI therapy is contraindicated. NB: NCCN ped ALL definitions used, ie BM 1% with confirmatory test AUTO-PY1 Phase 2 Expansion Inclusion:

i. CNS3 (treated to CNS2 eligible) ii. CNS pathology iii. Active of uncontrolled infection iv. Prior CD19-directed therapy (blinatumomab allowed) v. Grade ≥ 3 neurotoxicity following blinatumomab vi. <90 days from HSCT vii. Grade ≥ 2 acute GVHD or moderate/severe chronic GVHD requiring systemic steroids or other immunosuppressants within 4 weeks viii. Rapidly progressive disease or chemo-unresponsive disease NB: Blinatumomab cannot be used as bridging Obe-cel is being investigated as definitive therapy; therefore, HSCT is not part of treatment plan for this study Key Exclusion:

• AALL2523/AFFI2522: A Single-Arm, Open-Label, Multi-Centre, Phase 1b/2 Study Evaluating the Safety and Preliminary Efficacy of AUTO1 (Obe-cel) in Pediatric Patients with CD19-Positive Relapsed/Refractory (r/r) B cell Acute Lymphoblastic Leukemia (B ALL) and Aggressive Mature B cell NON-HODGKIN Lymphoma (B NHL) • Approved by SC August 5, 2025 • Approved by FDA October 1, 2025 • Activated in COG February 10, 2025 • Currently open at CHOP, Primary Children’s in Utah, and Methodist in San Antonio • Working on opening 8, additional COG sites – CHLA, Seattle, Cincinnati, Denver, CHOA, Lurie, Atlanta and Columbia Study Status

Acknowledgements • Lingyun Ji • Mignon Loh • Elizabeth Raetz • Maureen O’Brien • Nicole Towcimack • Megan Johnson • Deepa Bhojwani • Shannon Maude • Nirali Shah • Dave Teachey • Carrie Kitko • Mike Pulsipher • Lia Gore • Brent Wood GOSH • Sara Ghorashian • Persis Amrolia Autolus • Ram Malladi • Matthias Will • Ana Metelo • Michael Zhang • Pierre Lao-Sirieix

Autolus.com Q&A 46
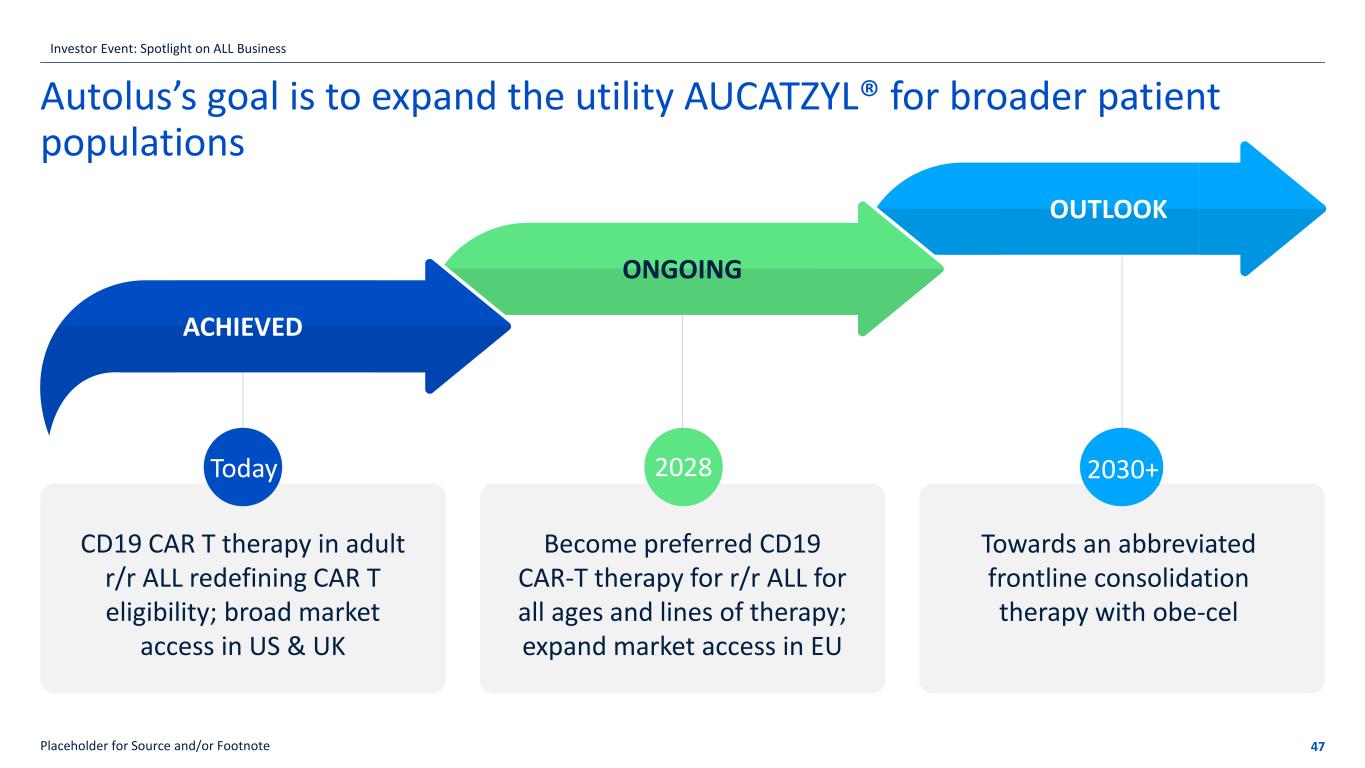
Autolus’s goal is to expand the utility AUCATZYL® for broader patient populations 47Placeholder for Source and/or Footnote CD19 CAR T therapy in adult r/r ALL redefining CAR T eligibility; broad market access in US & UK Become preferred CD19 CAR-T therapy for r/r ALL for all ages and lines of therapy; expand market access in EU Towards an abbreviated frontline consolidation therapy with obe-cel 2028 2030+ ACHIEVED ONGOING OUTLOOK Today Investor Event: Spotlight on ALL Business
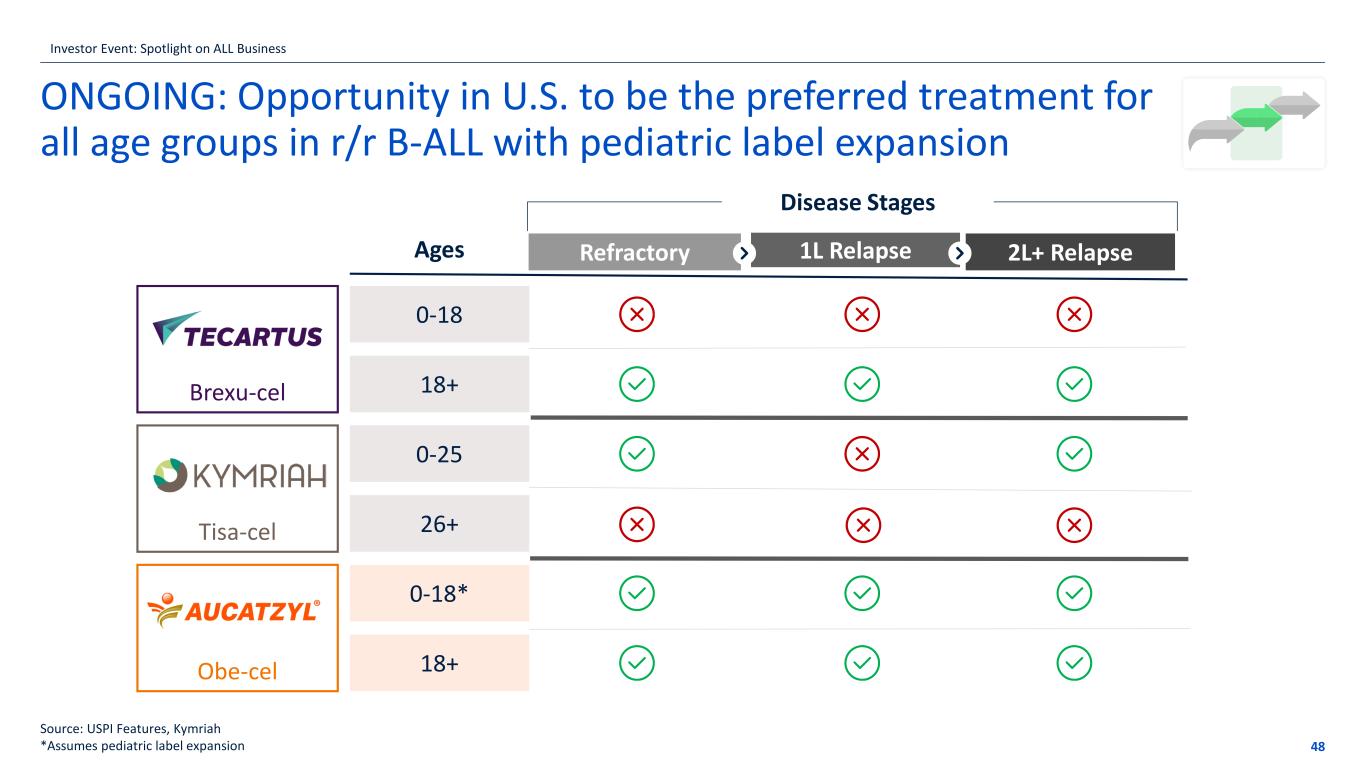
ONGOING: Opportunity in U.S. to be the preferred treatment for all age groups in r/r B-ALL with pediatric label expansion 48 Source: USPI Features, Kymriah *Assumes pediatric label expansion Brexu-cel Tisa-cel Obe-cel 0-18 18+ 0-25 26+ 0-18* 18+ Ages 1L RelapseRefractory 2L+ Relapse Disease Stages Investor Event: Spotlight on ALL Business
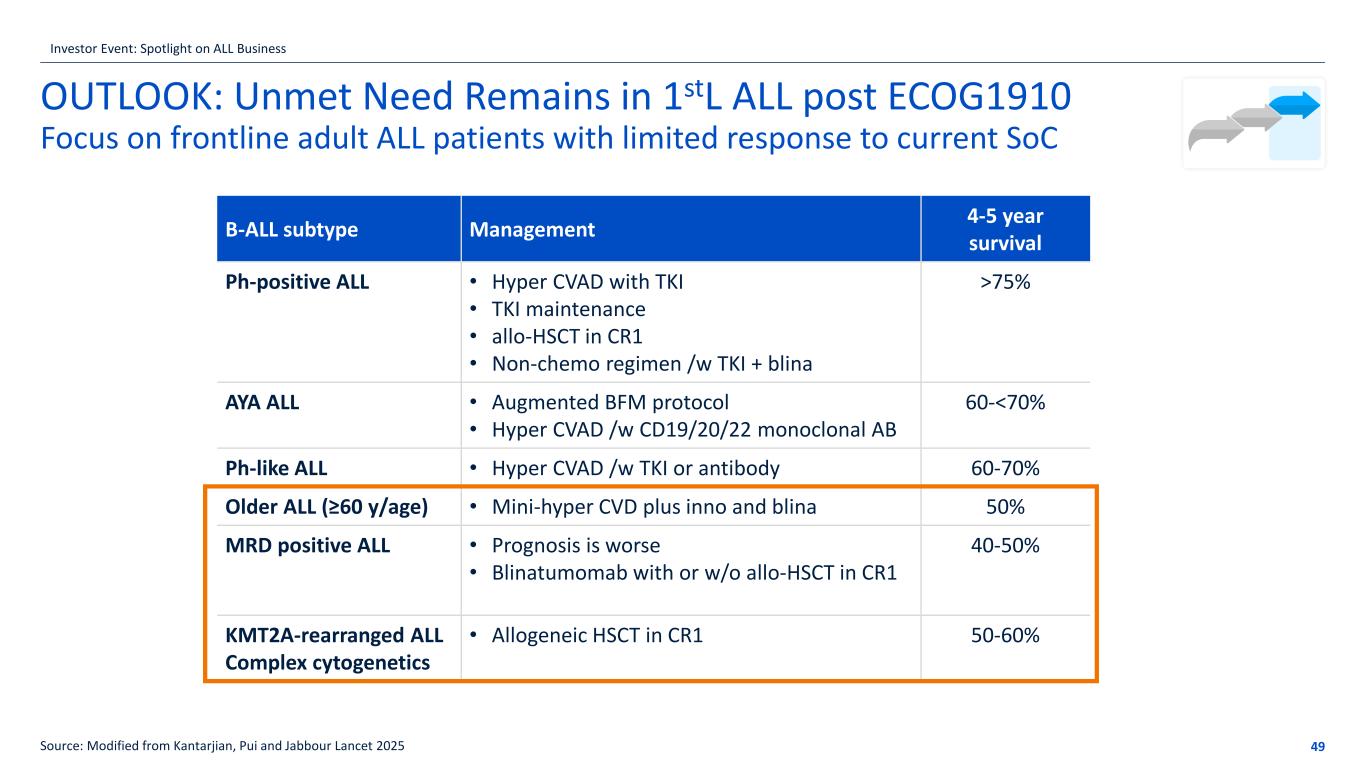
OUTLOOK: Unmet Need Remains in 1stL ALL post ECOG1910 Focus on frontline adult ALL patients with limited response to current SoC 49Source: Modified from Kantarjian, Pui and Jabbour Lancet 2025 B-ALL subtype Management 4-5 year survival Ph-positive ALL • Hyper CVAD with TKI • TKI maintenance • allo-HSCT in CR1 • Non-chemo regimen /w TKI + blina >75% AYA ALL • Augmented BFM protocol • Hyper CVAD /w CD19/20/22 monoclonal AB 60-<70% Ph-like ALL • Hyper CVAD /w TKI or antibody 60-70% Older ALL (≥60 y/age) • Mini-hyper CVD plus inno and blina 50% MRD positive ALL • Prognosis is worse • Blinatumomab with or w/o allo-HSCT in CR1 40-50% KMT2A-rearranged ALL Complex cytogenetics • Allogeneic HSCT in CR1 50-60% Investor Event: Spotlight on ALL Business
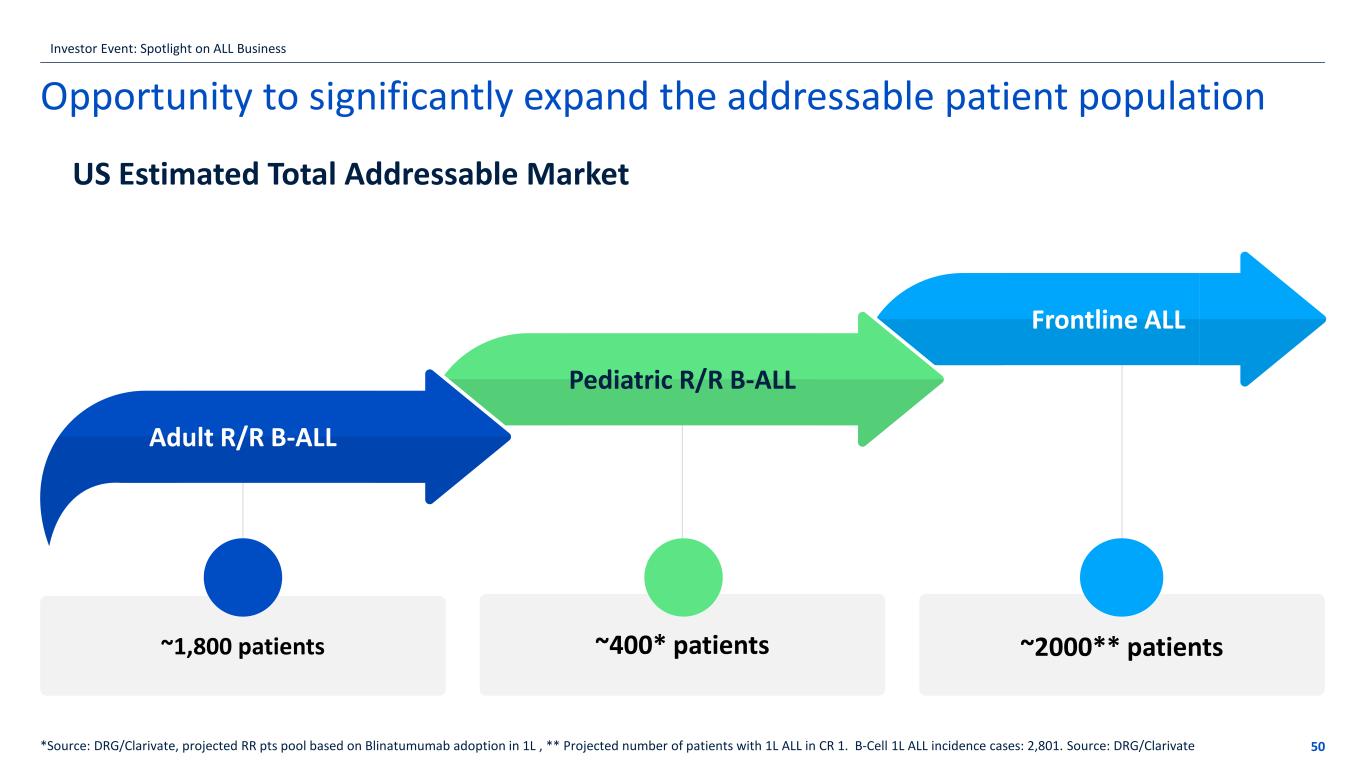
~1,800 patients Opportunity to significantly expand the addressable patient population 50*Source: DRG/Clarivate, projected RR pts pool based on Blinatumumab adoption in 1L , ** Projected number of patients with 1L ALL in CR 1. B-Cell 1L ALL incidence cases: 2,801. Source: DRG/Clarivate ~400* patients ~2000** patients Adult R/R B-ALL Pediatric R/R B-ALL Frontline ALL US Estimated Total Addressable Market Investor Event: Spotlight on ALL Business

Autolus.com Thank you 51
